Intracellular transport: This page is under construction!
Piecewise-linear approximation of cargo paths
We would like to infer the behavior of the molecular motors that transport the cargo, but because the motor heads are so small (~ 5 nm) they cannot be observed directly in live cells. Meanwhile, intracellular cargo can be quite large and “easily” visualized. The compromise is that diffusion is rapid and the observations are noisy compared to the speed of the walking motors.
Over the last decade graduate students Melanie Jensen and Linh Do and postdoc Keisha Cook have worked to develop a changepoint algorithm and inference protocol for automatically segmenting paths and reporting a “best-fit” piecewise linear approximation of cargo paths.
An animated version of lysosome path in the periphery of an A549 cell.
Black: “current” position of the lysosome.
Grayscale: “recent” positions, the lighter the farther in the past.
Red: the inferred position of the contact between the molecular motor and the microtubule.
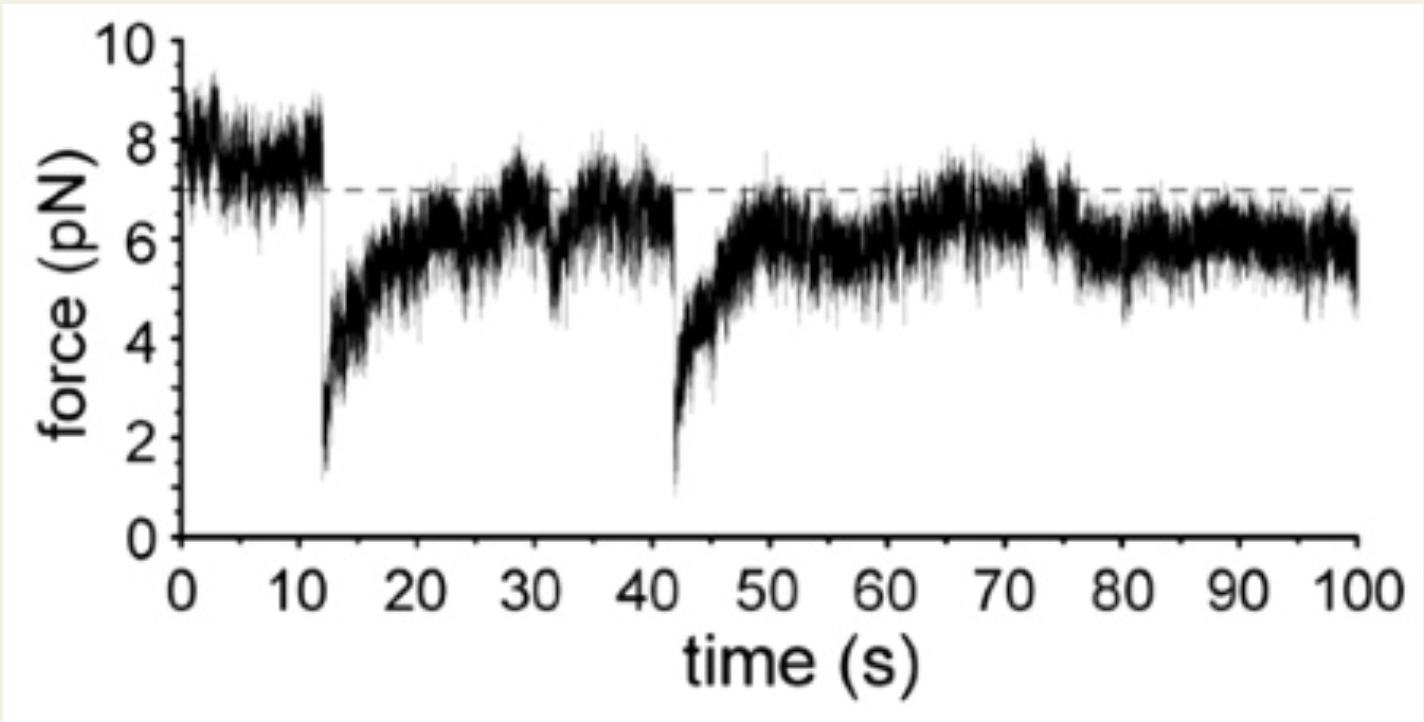
The Sysiphus experiments
Whether by optical traps or DNA tensiometers, experimentalists can explore molecular motor response to forces through “Sysiphus experiments.” In these experiments, an external force is applied to the cargo that a motor carries at a fixed location. After the motor binds to a nearby microtubule it processes away. As distance from the center of the trap increases, so does the force being translated to the motor. At some point the motor detaches, but this might be before or after reaching its critical “stall force”. The timing of detachment relative to the event of reaching stall force can be very informative. Eventually, the motor detaches and snaps back to the center of the force trap, only to rebind to the microtubule and start away towards its stall distance again.
In recent work with members of the Hancock Lab, we studied the behavior of kinesin-1, -2, and -3 motors in response to DNA tensiometers.
DNA tensiometer reveals catch-bond detachment kinetics of kinesin-1, -2 and -3. Crystal R Noell, Tzu-Chen Ma, Rui Jiang, Scott A McKinley, William O Hancock. Submitted November, 2025
Dynamic Instability and Microtubule Length Distributions
Minimal mechanisms of microtubule length distributions in living cells. Anna C Nelson, Melissa M Rolls, Maria-Veronica Ciocanel, and Scott A McKinley. Bulletin of Mathematical Biology. Vol 86, No 58, pgs 1-33. (2024)
Paper abstract: The microtubule cytoskeleton is responsible for sustained, long-range intracellular transport of mRNAs, proteins, and organelles in neurons. Neuronal microtubules must be stable enough to ensure reliable transport, but they also undergo dynamic instability, as their plus and minus ends continuously switch between growth and shrinking. This process allows for continuous rebuilding of the cytoskeleton and for flexibility in injury settings. Motivated by in vivo experimental data on microtubule behavior in Drosophila neurons, we propose a mathematical model of dendritic microtubule dynamics, with a focus on understanding microtubule length, velocity, and state-duration distributions. We find that limitations on microtubule growth phases are needed for realistic dynamics, but the type of limiting mechanism leads to qualitatively different responses to plausible experimental perturbations. We therefore propose and investigate two minimally-complex length-limiting factors: limitation due to resource (tubulin) constraints and limitation due to catastrophe of large-length microtubules. We combine simulations of a detailed stochastic model with steady-state analysis of a mean-field ordinary differential equations model to map out qualitatively distinct parameter regimes. This provides a basis for predicting changes in microtubule dynamics, tubulin allocation, and the turnover rate of tubulin within microtubules in different experimental environments.